
This experiment provided him with evidence that the majority of an atom’s mass was contained in the center of the atom. Rutherford, in an experiment that is now famously referred to as the Gold Foil Experiment, saw that while most alpha particles passed right through the gold foil, a few deflected to the side and some even bounced back. The atomic theory was further expanded upon in 1911 when Thomson’s colleague Ernest Rutherford began experimenting with alpha particles to probe the atomic structure of gold foil. Give two other examples in which science may be influenced by a researcher’s culture, background, or society. Can you think of any other common day example we could use to model Thomson’s atom? This is an example of how scientific knowledge might be subjective to the scientist’s background. THINK – For modern day scientists like yourself, plum pudding may not be a relevant comparison. In case you are not familiar, the picture to the below is of a plum pudding. He hypothesized that these particles were contained in positively charged atoms, much like plums in a traditional English plum pudding. Using this tool he discovered electrons, which are small particles with a negative electric charge. Thomson began experimenting with a cathode ray tube, which is shown in the diagram to the left. 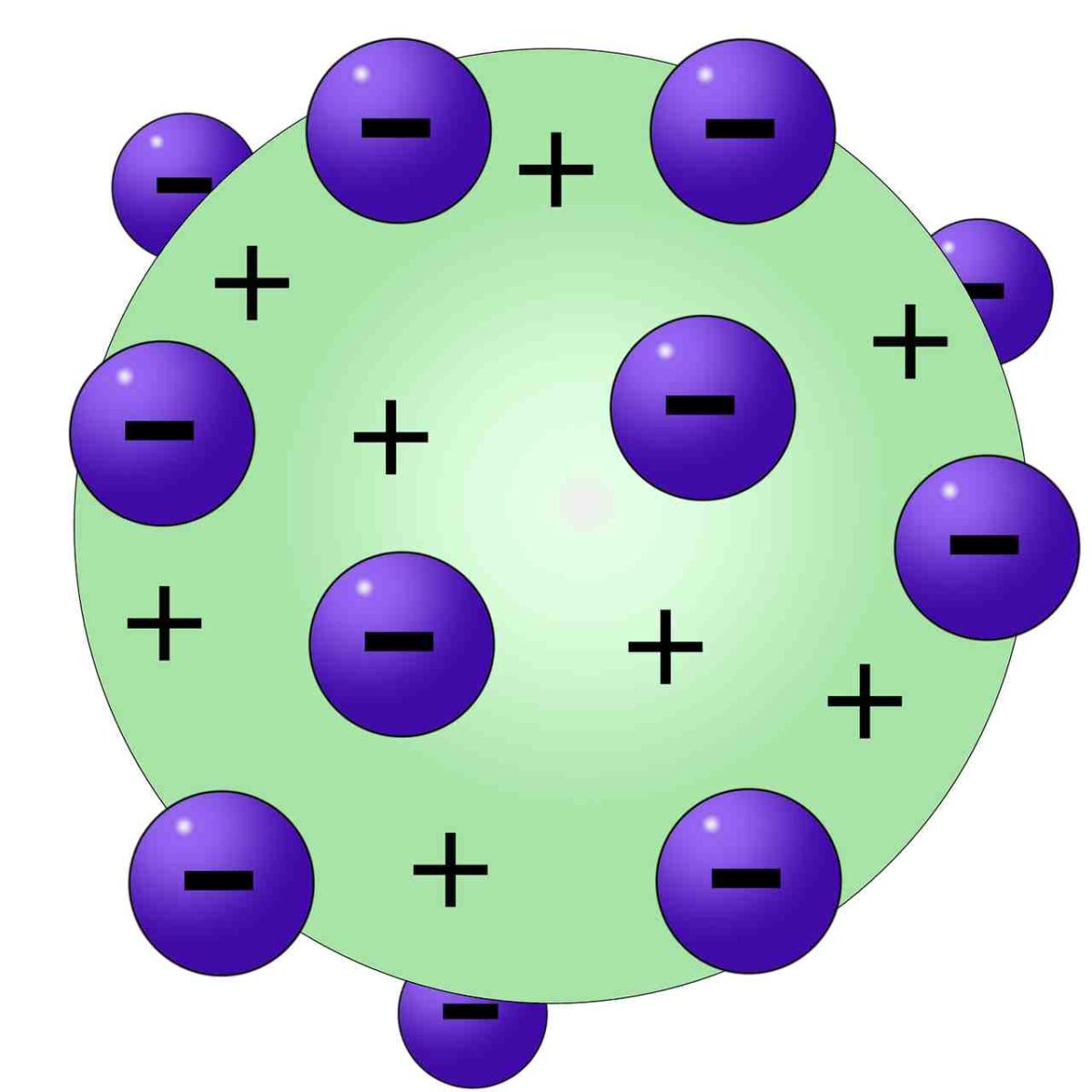
ThomsonĪn addition to Dalton’s atomic theory occurred in 1897 when English physicist J.J. What do you think were some of the reasons for this? Does the society we live in drive scientific progress or does scientific progress drive our societies? Explain your reasoning. THINK – It took over 2000 years to develop empirical evidence to support the atomic theory. He has also been memorialized in the chemistry field by the designation of the atomic mass unit as a Dalton. Since Dalton’s conclusions are based on an empirical analysis of experimental results, this is the first officially scientific atomic theory. Dalton’s work led him to develop the Law of Conservation of Mass (matter can neither be created nor destroyed) and Law of Constant Composition (elements combine in a fixed ratio to make up molecules). A chemical reaction results in the rearrangement of atoms in the reactant and product compounds.Compounds are produced through different whole-number combinations of atoms (“compounds” to be named “molecules” later).The atoms of different elements vary in size and mass.Everything is composed of atoms, which are indivisible building blocks of matter and cannot be destroyed.The Five Main Points of Dalton’s Atomic Theory This remained true until 1803, when an English chemist, John Dalton, came up with actual experimental evidence for the existence of atoms. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper.Maharshi Kanada’s atomic theory (or Democritus’s theory, depending on who you are reading) was largely a secondary interest for the majority of scientists since there was no evidence to support it. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. 
That is, samples that have the same mass ratio are not necessarily the same substance. 
It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
